Pharmaceutical intermediates are crucial building blocks in the complex process of drug manufacturing. Understanding their pharmaceutical intermediate definition is paramount for ensuring the quality, safety, and efficacy of medications worldwide. They represent a significant segment of the fine chemical industry, bridging the gap between basic raw materials and the final Active Pharmaceutical Ingredient (API). This understanding isn’t just for chemists and pharmaceutical professionals; it impacts global healthcare accessibility and affordability.
The global pharmaceutical intermediate market is a multi-billion dollar industry, projected to continue significant growth due to increasing demand for generic drugs and innovative therapies. Factors like stringent regulatory requirements, complex manufacturing processes, and the need for supply chain resilience drive the importance of reliable intermediate sourcing and production. Organizations like the UN and the World Bank recognize the vital role of a stable pharmaceutical supply chain in achieving global health objectives, particularly in developing nations.
Challenges within the industry, such as environmental concerns and cost pressures, highlight the need for advancements in sustainable manufacturing practices and innovative chemical processes. A thorough comprehension of pharmaceutical intermediate definition is thus not simply academic; it’s a practical necessity for navigating a dynamic and critically important industry.

The pharmaceutical industry is a cornerstone of modern healthcare, and pharmaceutical intermediates are the unsung heroes of drug production. The global market for these compounds is substantial, estimated at over $100 billion and projected to grow significantly in the coming years. This growth is driven by increasing demand for affordable medicines, especially generics, and the ongoing development of novel therapies. pharmaceutical intermediate definition, therefore, has widespread consequences for global health.
Supply chain vulnerabilities and geopolitical factors frequently disrupt pharmaceutical manufacturing, emphasizing the critical need for diversified sourcing of intermediates. The COVID-19 pandemic starkly illustrated these weaknesses, leading to shortages of essential medications. Robust, resilient supply chains for pharmaceutical intermediate definition are not merely commercial concerns, but are essential for national and global security.
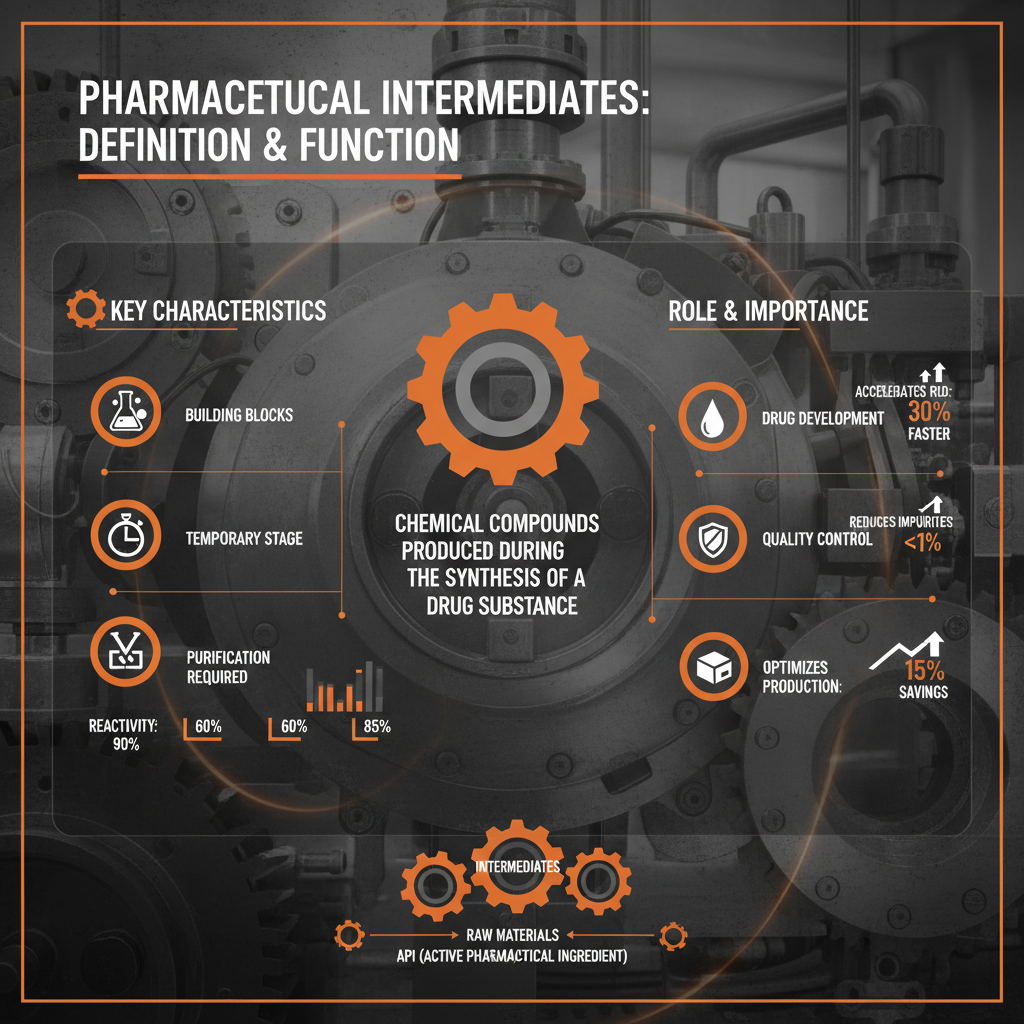
A pharmaceutical intermediate is a chemical compound formed during the synthesis of an Active Pharmaceutical Ingredient (API). It’s not the final drug product itself, but a precursor that undergoes further chemical transformations to become the API. Crucially, these intermediates must meet stringent quality standards, as impurities present at this stage can ultimately impact the safety and efficacy of the finished drug.
The connection to modern industry is undeniable. Pharmaceutical companies rarely manufacture APIs from basic raw materials. Instead, they rely on specialized intermediate manufacturers to produce these complex compounds. This division of labor allows for greater efficiency, cost-effectiveness, and access to specialized expertise. Understanding the pharmaceutical intermediate definition is essential for quality control, regulatory compliance, and innovation.
These intermediates serve a critical humanitarian need by enabling the affordable production of life-saving medications. Without a reliable supply of quality intermediates, access to essential drugs, especially in developing countries, would be severely compromised.
Several key factors define the quality and usability of pharmaceutical intermediates. Firstly, Purity is paramount. Impurities must be minimized and rigorously controlled to prevent adverse effects in the final drug product. Secondly, Consistency is vital. Batch-to-batch variations can disrupt downstream processing and affect API quality. Therefore, well-defined manufacturing processes are essential.
Thirdly, Scalability is crucial. The intermediate manufacturing process must be readily scalable to meet fluctuating demand, from small-scale clinical trials to large-scale commercial production. Fourthly, Cost-Effectiveness is a significant driver, particularly in the generics market. Efficient manufacturing processes and competitive sourcing of raw materials are essential.
Finally, Regulatory Compliance is non-negotiable. Intermediate manufacturers must adhere to stringent guidelines set by regulatory bodies like the FDA and EMA, including Good Manufacturing Practices (GMP). These factors collectively determine the suitability of an intermediate for pharmaceutical applications.
Pharmaceutical intermediates are universally applied across all therapeutic areas, from antibiotics and cardiovascular drugs to oncology and central nervous system medications. In regions like India and China, a significant portion of global intermediate production is concentrated, serving as key suppliers to pharmaceutical companies worldwide. These intermediates are utilized in the manufacturing of both branded and generic drugs.
The application extends to contract manufacturing organizations (CMOs) that specialize in API production on behalf of larger pharmaceutical companies. In post-disaster relief operations, the rapid production of essential medicines relies heavily on the availability of pre-manufactured intermediates. Remote industrial zones often benefit from the economic activity generated by intermediate production facilities.
Utilizing high-quality pharmaceutical intermediates offers numerous tangible benefits. Firstly, Cost Reduction is achieved through optimized manufacturing processes and reduced waste. Secondly, Sustainability is enhanced by employing greener chemistry principles and minimizing environmental impact.
Beyond the practical advantages, there's a significant Social Impact. Reliable access to affordable medications improves public health outcomes and reduces healthcare disparities. There is also the increased Reliability of supply chains due to robust quality control measures and diversified sourcing. These benefits build Trust with both patients and healthcare providers, fostering confidence in the pharmaceutical industry.
The future of pharmaceutical intermediate manufacturing is being shaped by several key trends. Continuous Manufacturing is gaining traction, offering improved efficiency and quality control compared to traditional batch processing. Digital Transformation, including the use of artificial intelligence and machine learning, is optimizing process parameters and predicting potential issues.
Green Chemistry principles are driving the development of more sustainable manufacturing routes, utilizing renewable feedstocks and minimizing hazardous waste. Increased focus on Automation and robotics is reducing manual labor and enhancing precision. These innovations collectively aim to create a more efficient, sustainable, and resilient pharmaceutical supply chain.
One of the primary challenges is the increasing complexity of API molecules, requiring more sophisticated and challenging intermediate syntheses. Another challenge is the need to reduce environmental impact, minimizing waste and hazardous materials. Furthermore, maintaining supply chain security and preventing counterfeiting are ongoing concerns.
Solutions include investing in research and development of innovative chemical processes, embracing green chemistry principles, and implementing robust traceability systems. Collaborative efforts between intermediate manufacturers, pharmaceutical companies, and regulatory agencies are also crucial for addressing these challenges. Advanced analytical techniques and real-time monitoring can enhance quality control and prevent deviations.
Finally, diversifying sourcing options and building strategic partnerships can mitigate supply chain risks and ensure a stable supply of critical pharmaceutical intermediate definition.
| Challenge | Impact on Production | Potential Solution | Implementation Cost (1-10) |
|---|---|---|---|
| Increasing API Complexity | Requires complex intermediate syntheses, lower yields | Investment in advanced chemical methodologies | 8 |
| Environmental Concerns | Waste generation, hazardous material usage | Adoption of Green Chemistry principles | 7 |
| Supply Chain Vulnerabilities | Disruptions, shortages, price fluctuations | Diversification of sourcing, strategic partnerships | 6 |
| Counterfeiting & Quality Control | Compromised drug safety & efficacy | Robust traceability systems, advanced analytics | 9 |
| Regulatory Compliance | Increased scrutiny, potential penalties | GMP adherence, continuous training | 7 |
| Scalability Challenges | Difficulty meeting fluctuating demand | Investment in flexible manufacturing technologies | 8 |
A pharmaceutical intermediate is a building block used to create an API (Active Pharmaceutical Ingredient). The API is the biologically active component in a drug that produces the intended effect. Intermediates undergo further chemical processing to become the API. Think of it like ingredients in a recipe versus the finished dish; intermediates are the ingredients, and the API is the final product.
The FDA doesn’t directly approve pharmaceutical intermediates, but they do regulate their manufacturing under Good Manufacturing Practices (GMP). Intermediate manufacturers must demonstrate that their processes consistently produce compounds of the required purity and quality. This is often assessed through audits and reviews of manufacturing records. The FDA focuses on ensuring that impurities are controlled to acceptable levels that won’t affect the safety of the final drug product.
Purity requirements for intermediates vary depending on the specific compound and its intended use in the API synthesis. However, generally, pharmaceutical intermediates need to have very high purity – often exceeding 98% or even 99%. The level of acceptable impurities is tightly controlled and specified in regulatory guidelines. Detailed analytical testing, like HPLC and GC-MS, are crucial for verifying purity.
Sustainability is becoming increasingly important. Manufacturers are focusing on adopting “Green Chemistry” principles – using renewable feedstocks, minimizing waste, and employing less hazardous solvents and reagents. Reducing the environmental impact of intermediate production is not only ethically responsible but also economically beneficial through lower waste disposal costs and improved resource efficiency.
Mitigation strategies include diversifying sourcing to multiple suppliers, building strategic partnerships with key intermediate manufacturers, and maintaining safety stock levels. Implementing robust supply chain monitoring systems and conducting regular risk assessments are also vital. Companies can also consider near-shoring or re-shoring production to reduce reliance on distant suppliers.
Technological advancements include continuous manufacturing processes, which offer improved efficiency and quality control, and the use of AI and machine learning for process optimization and predictive maintenance. Flow chemistry and microreactors are also gaining prominence, enabling more efficient and safer reactions. Automation and robotics are further streamlining production and reducing human error.
Understanding the pharmaceutical intermediate definition is fundamental to comprehending the complexities of drug manufacturing. These compounds are the essential building blocks that enable the production of life-saving medications, playing a critical role in global healthcare. Factors like purity, consistency, scalability, cost-effectiveness, and regulatory compliance are paramount for ensuring the quality and safety of pharmaceutical products.
Looking ahead, continued innovation in green chemistry, continuous manufacturing, and digital transformation will be crucial for creating a more sustainable, efficient, and resilient pharmaceutical supply chain. Investing in research, fostering collaboration, and prioritizing quality control will be essential for meeting the growing global demand for affordable and effective medications. Visit our website at www.kxdchem.com to learn more about our commitment to pharmaceutical intermediate solutions.




