Pharmaceutical intermediates are the crucial building blocks in the complex process of drug manufacturing. These compounds are formed during the synthesis of an Active Pharmaceutical Ingredient (API), but are not the API themselves. The global market for pharmaceutical intermediates is experiencing substantial growth, driven by increasing demand for generic drugs, rising healthcare expenditure, and the expansion of the pharmaceutical industry in emerging economies. Understanding the intricacies of sourcing, quality control, and regulatory compliance related to pharmaceutical intermediates importers is paramount for pharmaceutical companies worldwide.
The role of pharmaceutical intermediates importers is becoming increasingly critical as pharmaceutical manufacturers seek to optimize their supply chains and reduce costs. Global supply chains are complex, and relying on reliable partners for intermediates is essential to ensure consistent product quality and timely delivery. This demand is fueled by the growing need for affordable medications and the ongoing research and development of new therapies. Effective collaboration with importers specializing in these compounds is fundamental to the success of modern pharmaceutical production.
Ultimately, the efficient and reliable import of pharmaceutical intermediates directly impacts patient access to life-saving medications. A robust supply chain, facilitated by experienced pharmaceutical intermediates importers, is not simply a logistical concern; it is a humanitarian imperative. The ability to quickly and safely source these compounds is essential in responding to public health crises and meeting the evolving needs of a global population.

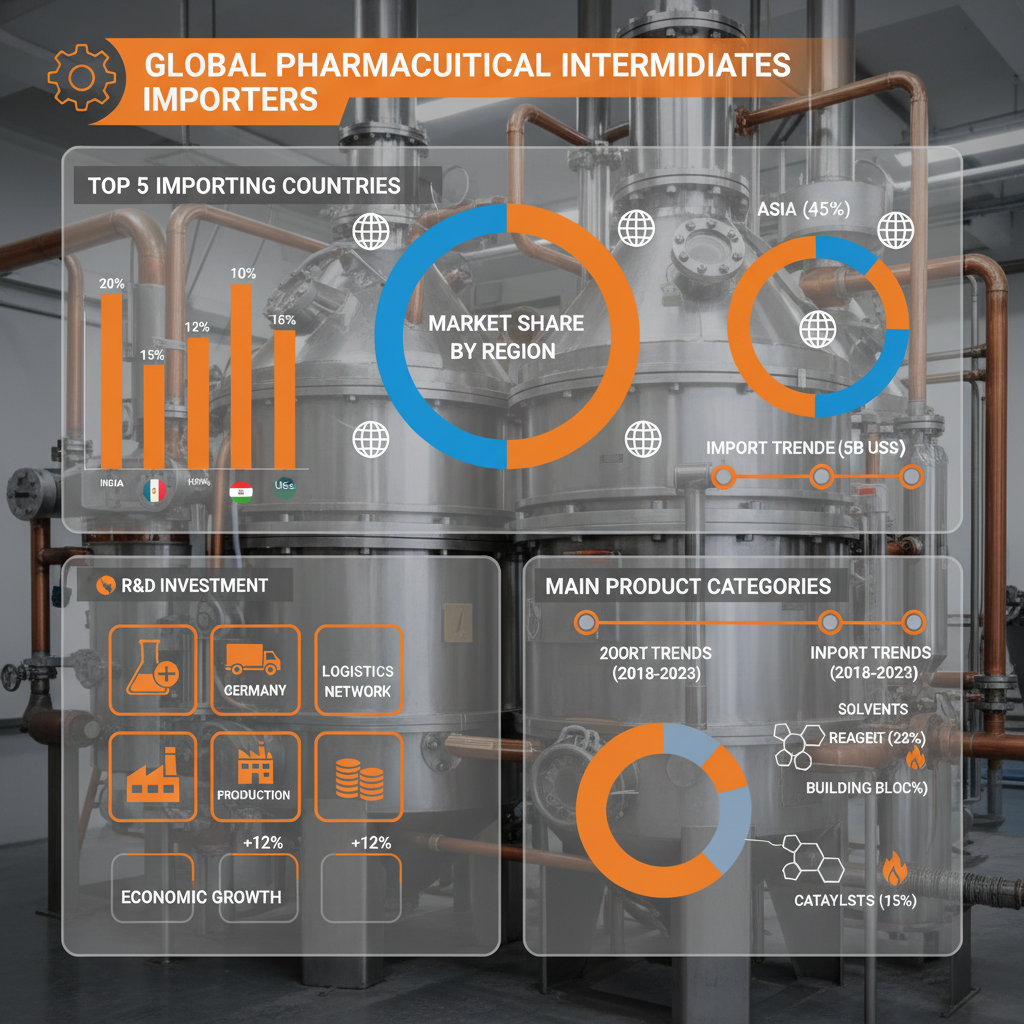
The pharmaceutical industry relies heavily on a complex network of suppliers, and pharmaceutical intermediates importers occupy a vital position within this ecosystem. They are responsible for sourcing, transporting, and ensuring the quality of crucial chemical compounds used in the synthesis of pharmaceuticals. Their expertise streamlines the supply chain, allowing pharmaceutical companies to focus on core competencies like research, development, and manufacturing of the final drug product.
These importers often specialize in specific types of intermediates or geographic regions, building strong relationships with manufacturers in countries like China, India, and Europe. This specialization allows them to navigate the regulatory landscape, manage logistics efficiently, and guarantee the authenticity and purity of the imported materials. Without their logistical prowess, many pharmaceutical production lines would face significant delays and disruptions.
Pharmaceutical intermediates importers are specialized businesses that facilitate the procurement of chemical compounds essential for the production of Active Pharmaceutical Ingredients (APIs). They act as a bridge between manufacturers of these intermediates – often located in countries with cost-effective production capabilities – and pharmaceutical companies requiring them for drug synthesis. They don't manufacture the intermediates themselves, but expertly manage the entire import process.
Their role extends beyond simple procurement. A reputable importer will provide services such as quality control testing, regulatory compliance documentation, logistical coordination, and often, just-in-time delivery. This ensures that pharmaceutical companies receive the necessary materials when they need them, meeting stringent quality standards and adhering to all relevant regulations.
Essentially, they provide a critical link in the pharmaceutical supply chain, contributing directly to the accessibility and affordability of medicines. The reliability and expertise of these importers directly impact the efficiency and cost-effectiveness of pharmaceutical production, ultimately influencing the global availability of essential drugs.
Several key factors define a successful pharmaceutical intermediates importer. First and foremost is a robust quality assurance system. This includes rigorous testing of all imported materials to verify purity, identity, and compliance with international standards like those set by the USP (United States Pharmacopeia) and EP (European Pharmacopoeia).
Secondly, regulatory expertise is paramount. Importers must be intimately familiar with the import regulations of both the exporting and importing countries, including documentation requirements, customs procedures, and any specific restrictions on certain chemicals. This prevents delays, ensures compliance, and minimizes the risk of legal issues.
Effective logistical management is also critical. This encompasses efficient transportation, secure storage, and timely delivery, often involving temperature-controlled shipping to maintain the integrity of sensitive compounds. Finally, strong supplier relationships are crucial for securing competitive pricing and ensuring a consistent supply of high-quality intermediates.
The global landscape for pharmaceutical intermediates importers is dynamic and influenced by several factors. Geopolitical instability, trade agreements, and currency fluctuations all impact sourcing strategies and pricing. China and India currently dominate the production of many key intermediates, offering cost advantages but also presenting risks related to quality control and regulatory compliance.
Recent global events have highlighted the vulnerability of concentrated supply chains. The COVID-19 pandemic, for example, caused significant disruptions to the flow of intermediates, leading to shortages and price increases. This has prompted many pharmaceutical companies to diversify their sourcing strategies and explore alternative suppliers.
While primarily serving the pharmaceutical industry, the expertise of pharmaceutical intermediates importers can extend to related sectors. Biotechnology companies, for instance, often require similar chemical compounds for research and development. Furthermore, some intermediates can find applications in the production of agrochemicals and specialized materials.
In post-disaster relief operations, rapid access to pharmaceutical ingredients is critical. Importers with established supply chains and logistical capabilities can play a vital role in quickly delivering essential medications to affected areas. This necessitates having robust contingency plans and relationships with transportation providers capable of operating in challenging environments.
Partnering with a dedicated pharmaceutical intermediates importer offers numerous benefits. Reduced costs through bulk purchasing and optimized logistics, minimized risk associated with regulatory compliance, and access to a wider range of suppliers are just a few examples.
Importantly, importers handle the complexities of international trade, allowing pharmaceutical companies to focus on their core business. They also provide valuable market intelligence, keeping clients informed about price trends, supply disruptions, and emerging regulatory changes.
The future of pharmaceutical intermediate importing will be shaped by several key trends. Increased digitalization of supply chains, leveraging technologies like blockchain to enhance transparency and traceability, is gaining momentum. The adoption of sustainable sourcing practices, emphasizing environmentally friendly manufacturing processes, is also becoming increasingly important.
Green chemistry principles are driving demand for intermediates produced using more sustainable methods, minimizing waste and reducing environmental impact. Furthermore, the rise of continuous manufacturing in pharmaceutical production will require importers to adapt to more frequent, smaller-volume deliveries, demanding greater agility and responsiveness.
Automation in warehousing and logistics, powered by AI and robotics, will further optimize efficiency and reduce costs. These innovations will require pharmaceutical intermediates importers to continually invest in new technologies and adapt their business models.
Despite the advancements, several challenges remain for pharmaceutical intermediates importers. Counterfeit intermediates pose a significant threat, requiring robust authentication procedures and collaboration with regulatory authorities. Maintaining consistent quality across diverse global suppliers is another persistent challenge, necessitating rigorous auditing and quality control programs.
Geopolitical risks and trade disputes can disrupt supply chains, requiring importers to diversify their sourcing strategies and build contingency plans. Addressing these challenges requires a proactive approach, embracing technologies like blockchain for traceability, investing in supplier audits, and fostering strong relationships with multiple suppliers.
The key to success lies in building resilient and adaptable supply chains, capable of withstanding unforeseen disruptions and maintaining a consistent flow of high-quality intermediates.
| Challenge Area | Severity (1-10) | Mitigation Strategy | Implementation Cost (USD) |
|---|---|---|---|
| Counterfeit Products | 9 | Blockchain Traceability, Supplier Audits | 50,000 |
| Quality Control Issues | 8 | Enhanced Testing Protocols, Independent Lab Verification | 30,000 |
| Supply Chain Disruptions | 7 | Diversified Supplier Base, Buffer Stock Management | 20,000 |
| Regulatory Compliance | 6 | Dedicated Regulatory Affairs Team, Continuous Training | 40,000 |
| Geopolitical Instability | 5 | Regional Sourcing, Political Risk Insurance | 15,000 |
| Logistics & Transportation | 4 | Strategic Carrier Partnerships, Real-time Tracking Systems | 10,000 |
Navigating international regulations is a complex process. Importers must comply with standards from organizations like the FDA (US), EMA (Europe), and local authorities. Documentation requirements for import permits, certificates of analysis, and country of origin statements are stringent. Furthermore, adherence to Good Distribution Practice (GDP) guidelines is essential to ensure product integrity throughout the supply chain. Failure to comply can lead to significant penalties and delays.
Implementing a robust quality assurance program is vital. This includes thorough supplier qualification audits, independent laboratory testing of each batch, and establishing clear specifications for purity and identity. It’s crucial to verify the supplier's manufacturing processes and ensure they adhere to relevant GMP (Good Manufacturing Practice) standards. A comprehensive documentation system and traceability protocols are also essential.
Documentation is paramount. Accurate and complete records are required for everything from sourcing and testing to transportation and storage. Key documents include Certificates of Analysis (COAs), Material Safety Data Sheets (MSDS), import permits, and invoices. Maintaining detailed records is essential for regulatory compliance and traceability, and facilitates efficient recall procedures if necessary.
Diversifying the supplier base is a key mitigation strategy. Maintaining relationships with multiple suppliers in different geographic locations reduces reliance on any single source. Holding buffer stocks of critical intermediates can provide a short-term cushion during disruptions. Developing contingency plans and utilizing real-time tracking systems to monitor shipments are also crucial elements of a robust risk management program.
Lead times vary significantly depending on the origin, destination, and mode of transportation. Typically, lead times range from 4 to 12 weeks. Air freight is faster but more expensive, while sea freight is more cost-effective but takes longer. Factors like customs clearance, regulatory inspections, and potential port congestion can also impact delivery schedules. Effective planning and communication with the importer are crucial for managing expectations.
Geopolitical instability, trade wars, and political sanctions can significantly disrupt supply chains. Tariffs, export restrictions, and political unrest can lead to price increases, delays, and even shortages. Importers must stay informed about geopolitical developments and proactively adjust their sourcing strategies to mitigate these risks. Developing alternative sourcing options and diversifying the supplier base are essential for resilience.
In conclusion, pharmaceutical intermediates importers are the unsung heroes of the pharmaceutical industry, playing a critical role in ensuring the availability of affordable and life-saving medications. Their expertise in sourcing, quality control, and regulatory compliance is invaluable, streamlining supply chains and mitigating risks. By understanding the complexities of this vital function, pharmaceutical companies can optimize their operations and ultimately improve patient outcomes.
Looking ahead, embracing digitalization, sustainability, and diversification will be key to navigating the evolving landscape of pharmaceutical intermediate importing. Investing in advanced technologies, building strong supplier relationships, and prioritizing ethical sourcing practices will be crucial for ensuring a resilient and responsible supply chain for the future. Visit our website at www.kxdchem.com to learn more about our services and how we can support your pharmaceutical sourcing needs.
