Drug product intermediate compounds represent a critical link in the pharmaceutical supply chain, bridging the gap between initial research and the finished medication reaching patients. These intermediates are the building blocks, carefully synthesized and refined compounds that undergo further processing to become the active pharmaceutical ingredient (API). Understanding the intricacies of drug product intermediate production, quality control, and sourcing is paramount to ensuring the safety, efficacy, and accessibility of vital medicines globally. The demand for high-quality intermediates is consistently increasing, driven by the expanding pharmaceutical industry and the growing need for innovative therapies.
The global pharmaceutical industry relies heavily on a robust and reliable supply of drug product intermediate materials. Production is concentrated in regions with established chemical manufacturing infrastructure, such as India, China, and Europe, although logistical complexities and geopolitical factors can introduce supply chain vulnerabilities. Regulatory scrutiny surrounding intermediate manufacturing is intensifying, with greater emphasis on traceability, process validation, and impurity profiling to meet stringent quality standards set by agencies like the FDA and EMA. This increasing focus on quality and security underscores the importance of trusted suppliers and robust quality management systems.
Ultimately, efficient and cost-effective drug product intermediate production translates to more affordable and accessible medications for patients worldwide. The continued advancement of synthetic chemistry, coupled with the implementation of sustainable manufacturing practices, will be vital in addressing the evolving challenges and opportunities within this crucial sector of the pharmaceutical industry.
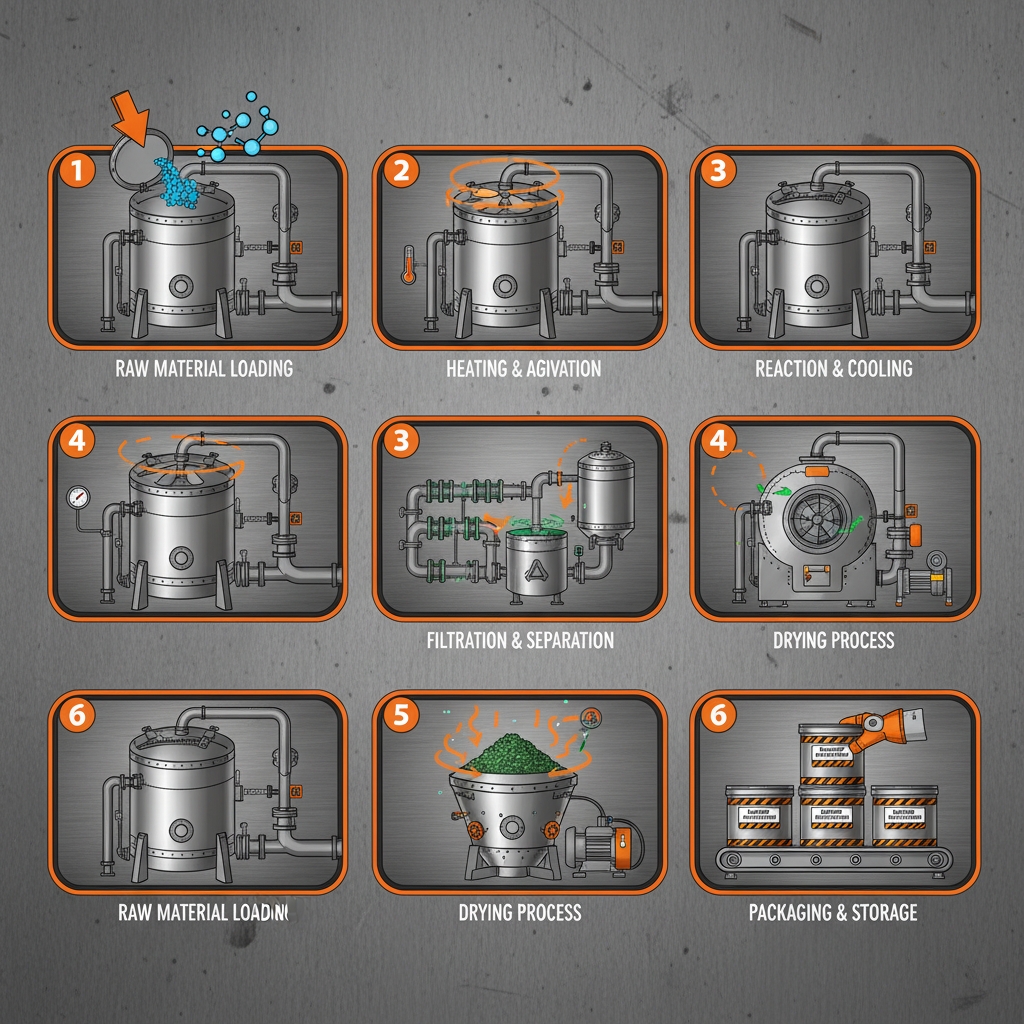

Drug product intermediate compounds are the crucial stepping stones in the complex journey of pharmaceutical manufacturing. These aren’t the final drug substance, but carefully crafted molecules that require further chemical transformation to achieve the desired therapeutic effect. Efficient production of these intermediates is directly linked to the cost and availability of essential medicines. drug product intermediate manufacturing demands meticulous attention to detail and adherence to strict quality standards.
The industry's dependence on these compounds necessitates a deep understanding of their chemical properties, synthetic routes, and potential impurities. Furthermore, sustainable and environmentally conscious manufacturing processes are increasingly important considerations, driving innovation in green chemistry and waste reduction techniques within the production of drug product intermediate.
The global market for pharmaceutical intermediates is estimated to be worth billions of dollars and continues to expand rapidly, driven by factors such as an aging global population, increasing prevalence of chronic diseases, and ongoing research and development efforts in the pharmaceutical sector. According to a recent report by the UN, the demand for affordable medicines is particularly acute in developing countries, highlighting the critical role of cost-effective drug product intermediate production.
Supply chain disruptions, as witnessed during the COVID-19 pandemic, have underscored the vulnerability of relying on a limited number of suppliers for key intermediates. This has prompted pharmaceutical companies to diversify their sourcing strategies and invest in regional manufacturing capabilities to enhance supply chain resilience. The World Bank emphasizes the importance of strengthening pharmaceutical manufacturing capacity in low- and middle-income countries to improve access to essential medicines.
A significant challenge is ensuring the quality and authenticity of drug product intermediate materials, particularly given the risk of counterfeit or substandard products entering the supply chain. International standards organizations, such as the ISO, are working to develop robust quality management systems and traceability protocols to mitigate these risks and safeguard patient safety.
A drug product intermediate is a chemical compound formed during the synthesis of an active pharmaceutical ingredient (API), but is not itself the API. It is essentially a “work in progress” molecule that requires additional chemical transformations to reach its final form. These intermediates can range from relatively simple molecules to complex structures, depending on the complexity of the API being manufactured.
The connection to modern industry and humanitarian needs lies in the fact that efficient production of these intermediates directly impacts the cost, availability, and quality of life-saving medications. Without a reliable supply of high-quality drug product intermediate, the entire pharmaceutical manufacturing process can be stalled, leading to drug shortages and increased healthcare costs.
Consider a vaccine production scenario. Multiple intermediates are needed to create the viral components, adjuvants, and stabilizers that comprise the final vaccine formulation. The quality and availability of these intermediates directly influence the speed and scale of vaccine rollout, especially during public health emergencies. drug product intermediate supply is crucial for global health security.
Several key factors dictate the success and efficiency of drug product intermediate production. Firstly, Process Optimization is essential, focusing on maximizing yield, minimizing waste, and reducing reaction times. Secondly, Quality Control is paramount, requiring rigorous analytical testing to ensure the purity, identity, and potency of each intermediate.
Cost-Effectiveness is another crucial factor, as pharmaceutical companies are constantly seeking to reduce manufacturing costs without compromising quality. Finally, Regulatory Compliance with agencies like the FDA and EMA is non-negotiable, necessitating meticulous documentation and adherence to Good Manufacturing Practices (GMP).
Drug product intermediate find widespread application across diverse pharmaceutical sub-sectors, including small molecule drugs, biologics, and generic medications. For example, in the production of antibiotics, specific intermediates are crucial for building the beta-lactam ring structure. In the realm of cancer therapeutics, complex intermediates are required for synthesizing targeted therapies.
These materials are utilized by pharmaceutical companies globally, with a significant concentration of manufacturing activity in India and China. However, increasing regulatory pressures and a desire for supply chain diversification are driving growth in intermediate production in regions like North America and Europe. In post-disaster relief operations, readily available intermediate stocks are vital for quickly ramping up production of essential medicines.
The strategic sourcing and efficient production of drug product intermediate offer significant advantages, including reduced manufacturing costs, improved supply chain security, and faster time-to-market for new drugs. Furthermore, investing in sustainable manufacturing processes can minimize environmental impact and enhance a company's reputation.
The long-term value lies in the ability to consistently deliver high-quality medications to patients, fostering trust and enhancing public health outcomes. Robust quality control and traceability measures contribute to patient safety and build confidence in the pharmaceutical industry. drug product intermediate quality is non-negotiable.
Several exciting trends are shaping the future of drug product intermediate production. Continuous flow chemistry, utilizing microreactors, offers improved process control and scalability. Biocatalysis, leveraging enzymes to catalyze chemical reactions, provides a more sustainable and environmentally friendly alternative to traditional chemical synthesis.
Digital transformation, incorporating automation, data analytics, and artificial intelligence, is optimizing manufacturing processes and improving quality control. Furthermore, increased focus on green chemistry principles – utilizing renewable feedstocks and minimizing waste – is driving innovation in sustainable manufacturing practices. The rise of personalized medicine also creates a demand for more specialized and customized drug product intermediate.
The integration of blockchain technology to enhance supply chain traceability and combat counterfeiting is also gaining momentum, providing greater transparency and security throughout the pharmaceutical supply chain.
The cost of drug product intermediate is influenced by several factors, including raw material prices, manufacturing complexity, regulatory compliance costs, and supply chain logistics. Global economic conditions and geopolitical events can also impact pricing. Utilizing efficient synthetic routes, optimizing process yields, and sourcing from reputable suppliers can help mitigate costs. Furthermore, economies of scale play a significant role, with larger production volumes generally leading to lower per-unit costs.
Quality control is absolutely critical in drug product intermediate manufacturing. Impurities or inconsistencies in intermediates can directly impact the quality, safety, and efficacy of the final drug product. Rigorous analytical testing, including chromatography, spectroscopy, and titration, is essential to ensure the purity, identity, and potency of each intermediate. Adherence to Good Manufacturing Practices (GMP) and stringent documentation protocols are also paramount.
Key regulatory requirements for drug product intermediate production include compliance with Good Manufacturing Practices (GMP) as outlined by agencies like the FDA and EMA. This encompasses process validation, impurity profiling, traceability, and comprehensive documentation. Manufacturers must also adhere to environmental regulations and safety standards. Regular audits and inspections are conducted to ensure compliance.
Pharmaceutical companies can mitigate supply chain risks by diversifying their sourcing strategies, establishing relationships with multiple suppliers, and investing in regional manufacturing capabilities. Maintaining buffer stocks of critical intermediates and implementing robust risk management plans are also essential. Supply chain visibility through technologies like blockchain can enhance transparency and traceability.
Sustainability is increasingly important in drug product intermediate production. This involves adopting green chemistry principles, utilizing renewable feedstocks, minimizing waste generation, and reducing energy consumption. Biocatalysis, continuous flow chemistry, and solvent recycling are examples of sustainable manufacturing practices. Companies are also focusing on reducing their carbon footprint and minimizing environmental impact.
Emerging technologies such as continuous flow chemistry, biocatalysis, digital twins, and artificial intelligence are significantly impacting drug product intermediate manufacturing. Continuous flow chemistry offers improved process control and scalability, while biocatalysis provides a more sustainable alternative to traditional chemical synthesis. AI-powered data analytics optimize processes and enhance quality control.
In conclusion, drug product intermediate represent a vital, yet often unseen, cornerstone of the pharmaceutical industry. Their consistent quality, availability, and cost-effectiveness directly impact patient access to life-saving medications. Embracing innovative technologies, prioritizing sustainability, and building resilient supply chains are critical for ensuring the continued success and integrity of this crucial sector.
Looking ahead, increased collaboration between pharmaceutical companies, intermediate manufacturers, and regulatory agencies will be essential to address emerging challenges and unlock new opportunities. Investing in research and development to develop more efficient and sustainable synthetic routes, along with implementing robust quality management systems, will further solidify the position of drug product intermediate as the foundation for a healthier future. Visit our website: www.kxdchem.com.




